 
Bohr assumed that the electrons moved in circular orbits around the nucleus. It was a modification of Rutherford's atomic model in which the atom is like "a microscopic solar system" in which the electrons orbit the nucleus. 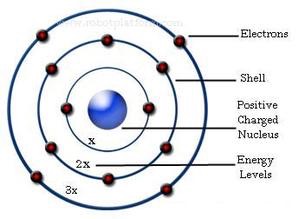
In 1913, Niels Bohr achieved worldwide celebrity in the field of Physics by publishing a series of essays in which he revealed his particular model of the structure of the atom. Borh, in a display of audacity that was unpredictable in his timid and withdrawn character, dared to sidestep these problems that hindered Rutherford's progress with a solution as simple as it was risky: he simply stated that the movements that occurred within the atom they are governed by laws foreign to those of traditional Physics. Rutherford had elaborated a theory of the atom that was totally valid on a speculative plane, but that could not be sustained within the laws of classical Physics. of the atom.įrom then on, a close collaboration was established between the two scientists that, sustained by firm ties of friendship, would be as lasting as it was fruitful. The doctoral thesis that the young Bohr had just read in Copenhagen was dedicated precisely to the study of electrons, and that he had taken to British territory with the hope of seeing it translated into English.īut Thomson was not enthusiastic about Bohr's work, so he decided to leave the Cavendish Laboratory and go to the University of Manchester, where he took advantage of the teachings of another Nobel laureate, Ernest Rutherford, to further his knowledge of radioactivity and models. Cambridge, under the tutelage of Sir Joseph John Thomson, a British chemist distinguished with the Nobel Prize in 1906 for his studies on the passage of electricity through the interior of gases, which had allowed him to discover the particle later baptized by Stoney as electron. After having revealed himself as a firm promise in the field of Nuclear Physics, he traveled to England to expand his knowledge at the Cavendish Laboratory of the University of Copenhagen. He studied Physics at the University of Copenhagen, where he obtained his doctorate in 1911. Son of Christian Bohr, a devout Lutheran, professor of physiology at the University of the city, and Ellen Adler, from a wealthy Jewish family of great importance in Danish banking, and in the "circles of Parliament".Ĭonsidered one of the most dazzling figures in contemporary Physics and, for his theoretical contributions and practical work, as one of the fathers of the atomic bomb, he was awarded the Nobel Prize in Physics in 1922 "for his research on the structure of atoms and the radiation emanating from them. He was involved with the establishment of CERN and the Research Establishment Risø of the Danish Atomic Energy Commission and became the first chairman of the Nordic Institute for Theoretical Physics in 1957.Danish physicist who made important contributions to the understanding of the structure of the atom and quantum mechanics. After the war, Bohr called for international cooperation on nuclear energy. From there, he was flown to Britain, where he joined the British Tube Alloys nuclear weapons project, and was part of the British mission to the Manhattan Project. In September 1943, word reached Bohr that he was about to be arrested by the Germans, and he fled to Sweden. After Denmark was occupied by the Germans, he had a famous meeting with Heisenberg, who had become the head of the German nuclear weapon project. Later, the element bohrium was named after him.ĭuring the 1930s, Bohr helped refugees from Nazism. 
He predicted the existence of a new zirconium-like element, which was named hafnium, after the Latin name for Copenhagen, where it was discovered. Bohr mentored and collaborated with physicists including Hans Kramers, Oskar Klein, George de Hevesy, and Werner Heisenberg. The notion of complementarity dominated Bohr's thinking in both science and philosophy.īohr founded the Institute of Theoretical Physics at the University of Copenhagen, now known as the Niels Bohr Institute, which opened in 1920. 
He conceived the principle of complementarity: that items could be separately analysed in terms of contradictory properties, like behaving as a wave or a stream of particles. Although the Bohr model has been supplanted by other models, its underlying principles remain valid. Bohr was also a philosopher and a promoter of scientific research.īohr developed the Bohr model of the atom, in which he proposed that energy levels of electrons are discrete and that the electrons revolve in stable orbits around the atomic nucleus but can jump from one energy level (or orbit) to another. "Niels Henrik David Bohr was a Danish physicist who made foundational contributions to understanding atomic structure and quantum theory, for which he received the Nobel Prize in Physics in 1922. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
